
From bench to bedside – and beyond: what we learned from our Academy of Medical Sciences North of England Cross‑Sector Hub launch event
The launch of our Academy of Medical Sciences’ North of England Cross‑Sector Hub
This week, we welcomed colleagues from academia, industry, the NHS, and regulatory experts to Newcastle for the launch of our Academy of Medical Sciences North of England Cross‑Sector Hub, hosted by the Northern Health Science Alliance (NHSA).
This first of three scheduled networking events focused on a topic where the North of England has exceptional strengths, but also well‑recognised challenges in translating and commercialising academic research in cell and gene therapies, for patient benefit.
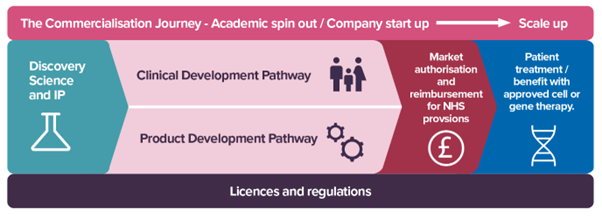
What followed was an open, honest and practical discussion about what it really takes to translate advanced therapies, not just into first‑in‑human trials, but into treatments that patients can access, health systems can deliver, and society can sustain.
Starting with the end in mind
A consistent message throughout the day was the importance of starting with the end in mind. As Professor Mimoun Azzouz, co-founder of two successful spin-out companies from the University of Sheffield, Blackfin Bio and Crucible Therapeutics., set out in his opening keynote, the translational pathway for advanced therapies is rarely linear. Even when the science is strong, there are two critical “valleys of death”:
- moving from bench to bedside, and
- moving from bedside to real‑world access and uptake

Professor Mimoun Azzouz
Scientific success alone is not enough. Manufacturing, regulatory strategy, clinical feasibility, affordability, and patient need all shape whether a therapy will ultimately make a difference. Importantly, the ‘end of the journey’ does not mean first‑in‑human trials, and it doesn’t even stop at market authorisation. True success is seen only when therapies reach patients in ways that are equitable, deliverable, and valued.
The value of early conversations
Across every session, from our fireside chat with Early Career Researchers (ECRs), to our interactive advisory clinic with a panel of experts, the same recommendation surfaced time and again: talk earlier, and to the right people. This means:
- engage manufacturers early, to avoid non‑translatable processes,
- speak with clinicians about how therapies would realistically be delivered,
- talk to regulators before problems arise,
- involve patients well before trial design.
Early conversations consistently de‑risk later failure. The message from colleagues at the Medicines and Healthcare Products Regulatory Agency (MHRA), the Advanced Therapy Treatment Centre (ATTC) network, and regional good manufacturing practice (GMP) and bioprocessing facilities was clear, regulatory and system challenges are often manageable, if addressed early.

Interactive advisory clinic on commercialisation
The North of England’s ecosystem advantage
One of the strongest take home messages from the day was just how complete the North of England’s advanced therapies ecosystem is, when convened at pan-northern scale. Across the North, we have:
- NHS centres delivering significant numbers of commercial cell and gene therapy trials
- academic‑embedded GMP manufacturing facilities supporting early translation
- process development and biomanufacturing innovation capabilities
- deep patient populations keen to engage in research
- a growing skills and training pipeline spanning students, technicians, clinicians, and researchers
Individually, these assets are powerful. Collectively, they form a system that can support innovators at every stage, if they know how to navigate it. That is exactly where the North of England Cross‑Sector Hub comes in.
Early career researchers at the centre
A particularly important part of the day was our fireside chat with ECRs from the British Society for Gene and Cell Therapy (BSGCT). Their insights were candid and sobering. Many routes into industry, regulation or clinical exposure still rely heavily on personal networks or chance opportunities. Time pressures, job insecurity, and the sheer breadth of skills expected of ECRs remain real barriers.

ECR fireside chat
But the conversation also highlighted solutions: mentoring, visibility, cross-sector mobility, and better alignment between academic training and translational realities. These are priorities we will continue to address, because the future of advanced therapies research and innovation in the North of England, and indeed the whole UK, depends on it.
Patients, access and affordability
Several of our expert contributors challenged the audience to push their thinking further still. Market authorisation does not guarantee patient access, and high‑cost therapies raise uncomfortable but necessary questions about affordability and value.
Patient and Public Involvement and Engagement (PPIE) was framed not as a procedural requirement, but as a strategic necessity, helping to shape endpoints, inform trial design, support investment cases, and ultimately ensure therapies address real unmet needs. If patients don’t want, or can’t access a therapy, innovators must ask hard questions, early, about whether they are solving the right problem.
A cross-sector Hub that is a platform, not a programme
The North of England Cross‑Sector Hub is not a finished offer. As we emphasised on the day, it is a platform, not a programme. Its success depends on continued participation from across the system to propose ideas, shape themes, host conversations, and test solutions together. Whether your role is in discovery science, manufacturing, clinical delivery, policy, or patient advocacy, there is a place for you in our network.
The strength of the North has always been its ability to collaborate at scale. This Hub is about embedding that way of working, not just for advanced therapies, but for the long‑term health and life sciences strength of our region.
Thank you to everyone who contributed so openly, our speakers, panellists, early career researchers, and delegates, and to colleagues at the Academy of Medical Sciences for such a constructive partnership.
Our speakers:
- Hannah Davies, Chief Executive Officer, Northern Health Science Alliance
- Rosalind Campion, Chief Executive Officer, Academy of Medical Sciences
- Professor Mimoun Azzouz, Chair of Translational Neuroscience, University of Sheffield and Co-founder, BlackfinBio and Crucible Therapeutics
- Dr Ellie Chilcott, Senior Research Fellow, EGA Institute for Women’s Health, University College London
- Dr Riccardo Privolizzi, Senior Research Fellow in the Genetic Therapy Accelerator Centre, UCL Queen Square Institute of Neurology
- Dr Alicia Teijeira Crespo, postdoctoral researcher, Cardiff University
- Dr Zoe Hewitt, Co-Founder and CEO, Regenerative Cell Therapy Consulting (Regen CTC)
- Dr Mark Jarvis, Director of Newcastle Advanced Therapies Dr Joe Cartwright, Head of Projects, Gene Therapy Innovation and Manufacturing Centre
(GTIMC), Sheffield - Professor Safwan Akram, Professor of Biomanufacturing, Engineering Biology and Innovation in Biosciences, National Horizons Centre, Teesside
- Prof. Fiona Thistlethwaite, Director, Innovate Manchester Advanced Therapy Centre Hub (iMATCH) and Advanced Therapy Treatment Centres (ATTC) network, Manchester
- Dr Tia Papoutselou, Clinical Development Scientist and Patient and Public Involvement Lead, Rinri Therapeutics
- Dr Andrew Zambanini, Head of Innovative Products – Innovation Accelerator and Regulatory Science; Medicines & Healthcare products Regulatory Agency
Our next Academy of Medical Sciences event




